In vitro -diagnostiikka (IVD)
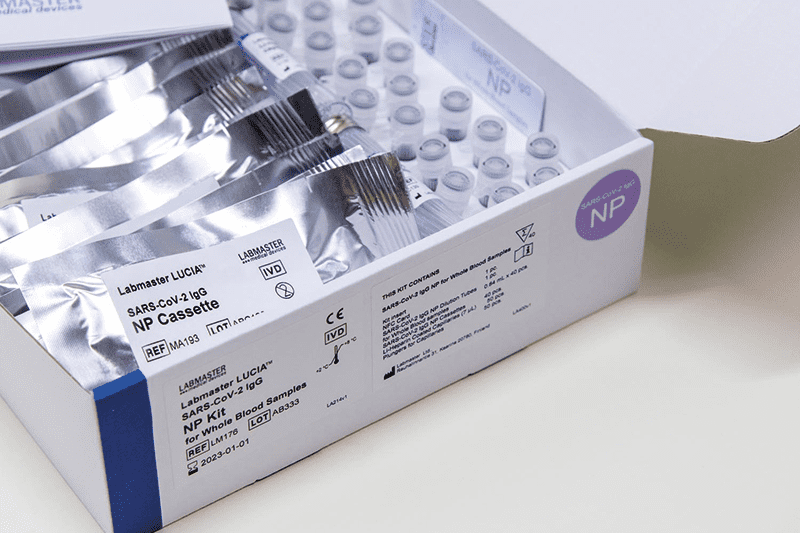
Labmaster LUCIA™ SARS-CoV-2 IgG NP -testipakkaus kokoverinäytteille
Ohjevideo: SARS-CoV-2 IgG NP -testi

info(at)labmaster.fi
Puh. +358 22 760 555
Rauhalinnantie 31, 20780 KAARINA, Finland
Legal disclaimer
Products designed, developed and manufactured by Labmaster Ltd. meet their specifications when stored, used and transported according to the instructions for use and product inserts. Labmaster LUCIA™ is a trademark designed and owned by Labmaster Ltd.
Not all products are available in all regions. Check with your local representative for availability in specific markets.

